EP0915812B1 - Biosoluble pot and marble (flame attenuated)-derived fiberglass - Google Patents
Biosoluble pot and marble (flame attenuated)-derived fiberglass Download PDFInfo
- Publication number
- EP0915812B1 EP0915812B1 EP98913041A EP98913041A EP0915812B1 EP 0915812 B1 EP0915812 B1 EP 0915812B1 EP 98913041 A EP98913041 A EP 98913041A EP 98913041 A EP98913041 A EP 98913041A EP 0915812 B1 EP0915812 B1 EP 0915812B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- metal oxide
- glass
- fibers
- sio
- sum
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Revoked
Links
- 239000004579 marble Substances 0.000 title claims description 22
- 230000002238 attenuated effect Effects 0.000 title claims description 4
- 239000011152 fibreglass Substances 0.000 title description 7
- 239000011521 glass Substances 0.000 claims description 60
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims description 43
- 239000000203 mixture Substances 0.000 claims description 39
- 239000000835 fiber Substances 0.000 claims description 34
- 239000002253 acid Substances 0.000 claims description 30
- 239000003365 glass fiber Substances 0.000 claims description 30
- 229910011255 B2O3 Inorganic materials 0.000 claims description 23
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims description 23
- 229910052593 corundum Inorganic materials 0.000 claims description 23
- 229910001845 yogo sapphire Inorganic materials 0.000 claims description 23
- 229910052681 coesite Inorganic materials 0.000 claims description 20
- 229910052906 cristobalite Inorganic materials 0.000 claims description 20
- 239000000377 silicon dioxide Substances 0.000 claims description 20
- 229910052682 stishovite Inorganic materials 0.000 claims description 20
- 229910052905 tridymite Inorganic materials 0.000 claims description 20
- 229910000272 alkali metal oxide Inorganic materials 0.000 claims description 19
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 claims description 11
- 239000000126 substance Substances 0.000 claims description 11
- KKCBUQHMOMHUOY-UHFFFAOYSA-N Na2O Inorganic materials [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 claims description 7
- 230000001747 exhibiting effect Effects 0.000 claims 4
- 239000007788 liquid Substances 0.000 claims 2
- 229910044991 metal oxide Inorganic materials 0.000 claims 2
- 150000004706 metal oxides Chemical class 0.000 claims 2
- 238000000034 method Methods 0.000 description 25
- 230000000052 comparative effect Effects 0.000 description 14
- 239000012530 fluid Substances 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 238000009413 insulation Methods 0.000 description 5
- 238000004090 dissolution Methods 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 239000006060 molten glass Substances 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 238000002425 crystallisation Methods 0.000 description 3
- 230000008025 crystallization Effects 0.000 description 3
- 239000004615 ingredient Substances 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 238000009616 inductively coupled plasma Methods 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000004417 polycarbonate Substances 0.000 description 2
- 229920000515 polycarbonate Polymers 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- IJALWSVNUBBQRA-UHFFFAOYSA-N 4-Isopropyl-3-methylphenol Chemical compound CC(C)C1=CC=C(O)C=C1C IJALWSVNUBBQRA-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 238000005452 bending Methods 0.000 description 1
- 239000013060 biological fluid Substances 0.000 description 1
- 210000004027 cell Anatomy 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000009795 derivation Methods 0.000 description 1
- 238000000921 elemental analysis Methods 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 210000003722 extracellular fluid Anatomy 0.000 description 1
- 238000007380 fibre production Methods 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 238000007496 glass forming Methods 0.000 description 1
- 239000011491 glass wool Substances 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 239000013067 intermediate product Substances 0.000 description 1
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N iron oxide Inorganic materials [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 1
- 235000013980 iron oxide Nutrition 0.000 description 1
- VBMVTYDPPZVILR-UHFFFAOYSA-N iron(2+);oxygen(2-) Chemical class [O-2].[Fe+2] VBMVTYDPPZVILR-UHFFFAOYSA-N 0.000 description 1
- 238000002386 leaching Methods 0.000 description 1
- 229910000464 lead oxide Inorganic materials 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 239000011160 polymer matrix composite Substances 0.000 description 1
- 229920013657 polymer matrix composite Polymers 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002787 reinforcement Effects 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C13/00—Fibre or filament compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/15203—Properties of the article, e.g. stiffness or absorbency
- A61F13/15252—Properties of the article, e.g. stiffness or absorbency compostable or biodegradable
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F13/00—Bandages or dressings; Absorbent pads
- A61F13/15—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators
- A61F13/53—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium
- A61F2013/530131—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium being made in fibre but being not pulp
- A61F2013/530328—Absorbent pads, e.g. sanitary towels, swabs or tampons for external or internal application to the body; Supporting or fastening means therefor; Tampon applicators characterised by the absorbing medium being made in fibre but being not pulp being mineral fibres, e.g. glass
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03C—CHEMICAL COMPOSITION OF GLASSES, GLAZES OR VITREOUS ENAMELS; SURFACE TREATMENT OF GLASS; SURFACE TREATMENT OF FIBRES OR FILAMENTS MADE FROM GLASS, MINERALS OR SLAGS; JOINING GLASS TO GLASS OR OTHER MATERIALS
- C03C2213/00—Glass fibres or filaments
- C03C2213/02—Biodegradable glass fibres
Definitions
- the present invention pertains to fiberglass products prepared from glass compositions suitable for fiberization by the pot and marble process.
- the glass fibers exhibit enhanced biosolubility while maintaining other desirable properties.
- Fiberglass has a myriad of uses, including the reinforcement of polymer matrix composites; preparation of thermoformable intermediate products for use as headliners and hoodliners in vehicles; air and water filtration media; and sound and thermal insulation products.
- the preparation and/or subsequent processing of such materials often involves handling steps which result in cut or broken fibers which may be inhaled.
- it has become important to create glass compositions which exhibit high degrees of biosolubility, i.e. which are rapidly solubilized in biological fluids.
- Glass fibers must also be strong and moisture-resistant. If moisture weakens glass fibers appreciably, their applicability to many uses suffers. Weakened glass fibers not only possess less than desired tensile strength and modulus, but also break and fracture more easily, thus increasing the risk of inhalation, etc. By the same token, moisture resistant glass fibers which have low strength to begin with also do not fulfill many requirements. For example, building insulation is shipped in compressed form. If the glass fibers of the insulation product are weak or brittle, many fibers will be broken during compression, not only increasing the number of small fibers which are bioavailable, but also producing an inferior product which may not recover a sufficient amount of its pre-compressed thickness. Strong fibers which are not moisture resistant also exhibit a great deal of breakage, especially under humid storage, as illustrated hereinafter. Finally, glass fibers must be prepared from glass compositions which can be economically processed.
- the two principle methods of glass wool fiber production are the pot and marble process and the centrifugal or "rotary" process.
- molten glass enters a centrifugal spinner from the forehearth of a glass melting furnace.
- centrifugal spinner rotates, relatively large diameter glass strands stream from orifices located in the spinner's periphery. These large diameter strands immediately contact an intense hot gas jet produced by burners located around the spinner.
- the hot gas attenuates the large diameter strands into fine, elongated fibers, which may be collected on a moving belt.
- the glass ingredients are first melted in a glass melter prior to their entry into the forehearth.
- the feed to the forehearth is high temperature, molten glass.
- the molten glass fed to the spinner is cooled to the HTV (high temperature viscosity) or "fiberization" temperature.
- the difference in temperature between the HTV and liquidus ( ⁇ T), the temperature which defines the boundary of crystallization, may be quite small in the rotary process.
- the primaries exiting the pot from the pot and marble process are flame attenuated rather than hot gas attenuated, thus exposing the glass fibers to higher temperatures than in the rotary process. These higher temperatures cause a loss of the more volatile compounds of the glass composition from the outside of the fibers, resulting in a "shell" which has a different composition than the fiber interior.
- the biosolubility of glass fibers prepared from pot and marble fiberglass is not the same as that derived from the rotary process.
- glass fibers must necessarily dissolve from the fiber ends or the cylindrical exterior, a more highly resistant shell will drastically impede the biodissolution. rate.
- glass fibers of enhanced biosolubility may be prepared from glass compositions suitable for pot and marble processing, which exhibit minimally about a 177°C (350°F) difference in HTV and liquidus, and which have well defined formulations meeting both narrow mol percentage composition as well as meeting each of three specific "C-ratios" which govern chemical resistance, moisture resistance, and biosolubility.
- the subject invention glasses have HTV and liquidus which are suitable for production of glass fibers in the pot and marble process.
- Such glass generally must have an HTV (10 3 poise) of 982°C (1800°F) to 1149°C (2100°F), preferably 1038°C (1900°F) to 1093°C (2000°F) and exhibit a liquidus which is minimally about 177°C (350°F) preferably 218°C (425°F), and more preferably 260°C (500°F) or more lower than the HTV.
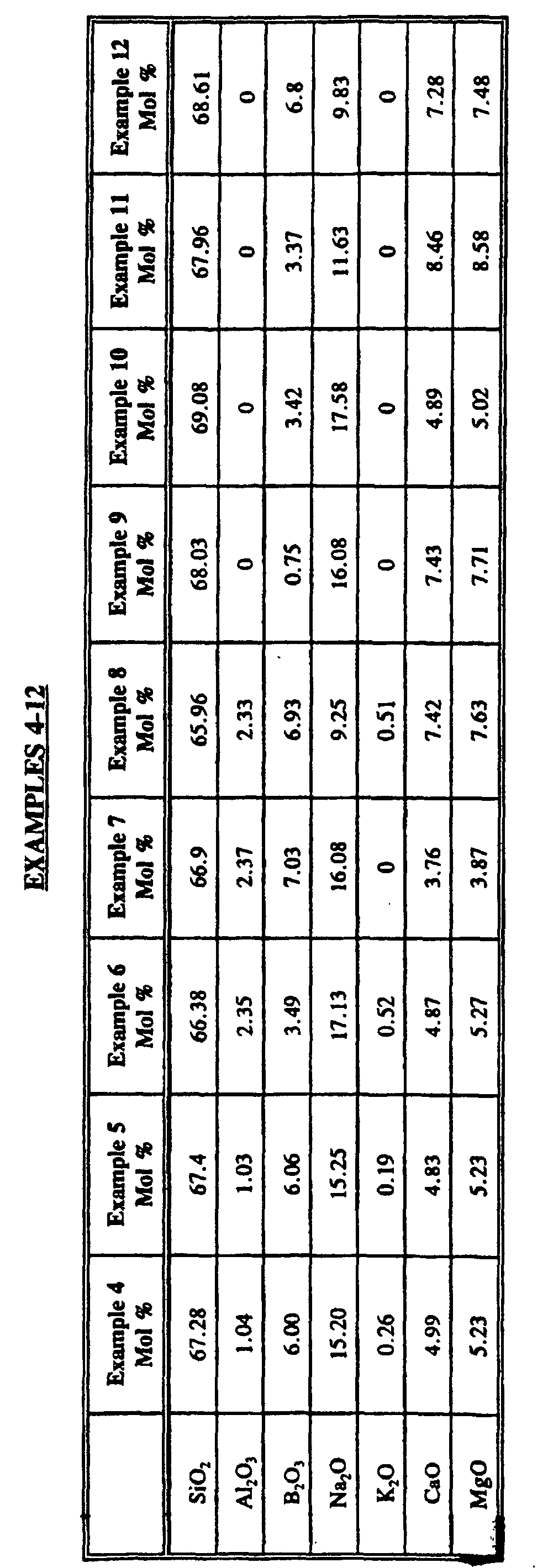
- the glass composition must fall within the_ following range of composition, in mol percent: SiO 2 65.96 -- 69.7 Al 2 O 3 0 -- 2.37 RO 7 -- 18 R 2 O 9 -- 20 B 2 O 3 0 -- 7.1
- R 2 O is an alkali metal oxide and RO is an alkaline earth metal oxide.
- R 2 O is preferably Na 2 O in most substantial part, while RO may be MgO and/or CaO, preferably both, in a molar ratio of MgO/CaO of 1:3 to 3:1, more preferably 2:3 to 3:2.
- the chemical behavior of the glass is dictated by three ratios which the glass composition must meet, C(acid), C(bio), and C(moist).
- C(acid) is the ratio which pertains to chemical resistance in acid environments
- C(bio) is the ratio which is most closely linked to biosolubility
- C(moist) is the ratio which relates to retention of properties in moist environments. It is desired that C(acid) and C(moist) be as large as possible, while C(bio) should be as low as possible.
- the HTV and liquidus of the overall composition must be suitable for glass fiber processing. It has been found that pot and marble glass of high biosolubility, while yet maintaining other necessary physical properties such as chemical resistance and moisture resistance, is obtained when C(acid) ⁇ 1.95, C(bio) ⁇ 2.30, and C(moist) ⁇ 2.40.
- the biosoluble fiberglass of the subject invention has a composition which falls within the following ranges (in mol percent): SiO 2 66 -- 69.0 Al 2 O 3 0 -- 2.2 RO 7 -- 16 R 2 O 9 -- 19 B 2 O 3 0 -- 7.1
- the biosoluble glass fibers of the subject invention have a composition which falls within the following most preferred range: SiO 2 66 -- 68.25 Al 2 O 3 0 -- 2.2 RO 7 -- 13 R 2 O 11 -- 18 B 2 O 3 0 -- 7.1
- C(acid) be greater than or equal to 2.00; C(bio) be less than or equal to 2.23, more preferably less than or equal to 2.20; and that C(moist) be greater than or equal to 2.50, preferably greater than or equal to 2.60.
- C(acid) and C(moist) be as high as possible.
- C(moist) values of 3.00 or greater are particularly preferred.
- the various C-ratios are independent in the sense that a more preferred glass need not have all "more preferred" C-ratios.
- Acid resistance may be measured by battery industry standard tests. For example, a typical test involves addition of 5 grams of nominally 3 ⁇ m diameter fiber in 50 mL of sulfuric acid having a specific gravity of 1.26. Following refluxing for 3 hours, the acid phase may be separated by filtration and analyzed for dissolved metals or other elements.
- the procedure used to evaluate biodissolution rate is similar to that described in Law et al. (1990).
- the procedure consists essentially of leaching a 0.5 gram aliquant of the candidate fibers in a synthetic physiological fluid, known as Gamble's fluid, or synthetic extracellular fluid (SEF) at a temperature of 37°C and a rate adjusted to achieve a ratio of flow rate to fiber surface area of 0.02 cm/hr to 0.04 cm/hr for a period of up to 1,000 hours duration.
- Fibers are held in a thin layer between 0.2 ⁇ m polycarbonate filter media backed by plastic support mesh and the entire assembly placed within a polycarbonate sample cell through which the fluid may be percolated.
- Fluid pH is regulated to 7.4 + 0.1 through the use of positive pressure of 5% CO 2 /95% N 2 throughout the flow system.
- Biodissolution rate constants in ng/cm 2 /hr are greater than 150 ng/cm 2 /hr, preferably greater than 200 ng/cm 2 /hr, more preferably greater than 300 ng/cm 2 /hr, and most preferably greater than 400 ng/cm 2 /hr.
- C-ratios are calculated for a conventional C-glass (chemically resistant glass) and a "soluble" glass as disclosed in Examples 1a and 2b in Table 1 of U.S. Patent No. 5,055,428.
- the glass composition is in weight percent. HTV (10 3 poise) and liquidus are as reported in the '428 patent.
- Comparative Example 1 (Wt%) Comparative Example 2 (Wt%) SiO 2 66.4 66.7 Al 2 O 3 1.2 1.0 B 2 O 3 11.0 10.0 Na 2 O 12.9 13.9 K 2 O 0.2 0.2 CaO 4.8 5.5 MgO 3.2 2.5 C(acid) 2.03 2.07 C(bio) 2.09 2.13 C(moist) 3.41 3.29 HTV (10 3 poise) 1073°C (1965°F) 1065°C (1949°F) Liquidus 948°C (1738°F) 928°C (1702°F)
- the C-ratios of these rotary process glasses indicate that they should both have good performance with respect to acid resistance, moisture resistance, and biosolubility.
- the Comparative Example 2 glass is reported by the patentee to have a dissolution rate in model physiological saline (composition not disclosed) of 211 ng/cm 2 /hr. However, examination of the HTV and liquidus temperatures reveals that these differ only by 126°C (227°F) and 137°C (247°F), respectively. Thus, these glass compositions cannot be used in pot and marble fiberization.
- These Comparative Examples serve to illustrate the ease with which higher biosolubility can be obtained in rotary processable glass. These glasses cannot be used to manufacture fiberglass by the pot and marble process. However, even were this possible, the flame attenuation and consequent loss of volatile oxides from the fiber surface would be expected to lower the measured biodissolution rate by a factor of from about 2 to 4.
- the C-ratios indicate that the glasses of Table 2 should exhibit desirable chemical resistance (both acid and moisture) as well as high biodissolution.
- the high biodissolution is confirmed by actual tests, being in both cases, considerably greater than 300 ng/cm 2 /hr.
- a subject invention glass is compared with two commercial glasses for acid resistance and moisture resistance, respectively.
- the formulations are as follows.
- Example 3 Example C3
- Example 3 The acid resistance of the Example 3 glass was compared with that of Comparative Example C3. It is noted that the Comparative Example C3 glass meets the C-ratio requirements but not the compositional limitations. The results of the acid resistance test are presented below in Table 3a. Glass Example 3 Example C3 Element Quantity Dissolved (ppm) Quantity Dissolved (ppm) Al 187 453 Ca 2831 4110 Mg 854 938 To determine moisture resistance, a stress corrosion test is used in which the fibers are stressed by bending in a controlled humidity and temperature test chamber. Fibers which exhibit moisture resistance under these conditions take longer to break. The Example 3 glass was compared to Comparative Example C4 glass, a glass used commercially for building insulation where compression of insulation and storage generates the potential for fiber breakage as a result. After 50 hours, only 12% of the Example 3 glass had broken, while all of the Comparative Example C4 fibers had failed.
- Comparative Example C4 glass a glass used commercially for building insulation where compression of insulation and storage generates the potential for fiber breakage
- C-ratios are calculated for the rotary process glasses of Example 3 of U.S. Patent No. 4,510,252, and Example 2 of U.S. Patent No. 4,628,038.
- Composition, calculated C-ratios, liquidus, and estimated HTV (10 3 poise) are given below in Table 4, in mol percent.
- Example C5 Example C6 SiO 2 68.2 66.67 Al 2 O 3 2.2 2.25 B 2 O 3 5.0 4.78 Na 2 O 9.2 8.68 K 2 O -- 0.26 CaO 11.9 14.77 MgO 3.5 3.60 C(acid) 1.85 1.94 C(bio) 2.03 2.15 C(moist) 2.54 2.70 HTV (10 3 poise), est. 1248°C (2280°F) 1210°C (2210°F) Liquidus 1084°C (1983°F) 1113°C (2035°F)
- the acid resistance of Comparative Example C5 is expected to be low, and the biodissolution is expected also to be low, although the glass should display good moisture resistance.
- the difference between HTV (10 3 poise) and liquidus is only about 147.2°C (297°F), and thus this glass is not suitable for use in a pot and marble process.
- the glass of Comparative Example C6 exhibits C(acid) close to an acceptable value, although C(bio) is too high.
- the glass should have good moisture resistance.
- the glass cannot be used in a pot and marble process as the difference between liquidus and HTV (10 3 poise) is only 79.4°C (175°F).
- Example 6 C-ratios and composition data (mol percent) are presented for Example 6 of U.S. Patent No. 5,108,957.
- the term “consisting essentially of” is meant that additional ingredients may be added provided they do not substantially alter the nature of the composition.
- Substances which cause the biodissolution to drop below 150 ng/cm 2 /hr or which lower the ⁇ T to a value below 177°C (350°F) are substances which do substantially alter the composition.
- the glass compositions are free of iron oxides, lead oxides, fluorine, phosphates (P 2 O 5 ), zirconia, and other expensive oxides, except as unavoidable impurities. It should be noted that while rotary process glass compositions are in general unsuitable for pot and marble fiberization, the reverse is not true, and the subject invention glass compositions should yield fibers prepared by the rotary process which have yet higher rates of biodissolution.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Glass Compositions (AREA)
- Filtering Materials (AREA)
Description
| SiO2 | 65.96 -- 69.7 |
| Al2O3 | 0 -- 2.37 |
| RO | 7 -- 18 |
| R2O | 9 -- 20 |
| B2O3 | 0 -- 7.1 |
| SiO2 | 66 -- 69.0 |
| Al2O3 | 0 -- 2.2 |
| RO | 7 -- 16 |
| R2O | 9 -- 19 |
| B2O3 | 0 -- 7.1 |
| SiO2 | 66 -- 68.25 |
| Al2O3 | 0 -- 2.2 |
| RO | 7 -- 13 |
| R2O | 11 -- 18 |
| B2O3 | 0 -- 7.1 |
| Comparative Example 1 (Wt%) | Comparative Example 2 (Wt%) | |
| SiO2 | 66.4 | 66.7 |
| Al2O3 | 1.2 | 1.0 |
| B2O3 | 11.0 | 10.0 |
| Na2O | 12.9 | 13.9 |
| K2O | 0.2 | 0.2 |
| CaO | 4.8 | 5.5 |
| MgO | 3.2 | 2.5 |
| C(acid) | 2.03 | 2.07 |
| C(bio) | 2.09 | 2.13 |
| C(moist) | 3.41 | 3.29 |
| HTV (103 poise) | 1073°C (1965°F) | 1065°C (1949°F) |
| Liquidus | 948°C (1738°F) | 928°C (1702°F) |
| Example 1 (mol %) | Example 2 (mol %) | |
| SiO2 | 67.24 | 67.18 |
| Al2O3 | 1.04 | 1.02 |
| B2O3 | 6.08 | 5.99 |
| CaO | 4.99 | 4.87 |
| MgO | 5.24 | 5.26 |
| Na2O | 15.22 | 15.45 |
| K2O | 0.26 | 0.23 |
| C(acid) | 2.05 | 2.05 |
| C(bio) | 2.15 | 2.14 |
| C(moist) | 2.89 | 2.87 |
| Biosol K(dis) | 350 | 426 |
| HTV | 1972 | 1981 |
| Liquidus | 1435 | < 1325 |
| Example 3 | Example C3 | Example C4 | |
| SiO2 | 67.28 | 65.36 | 57.53 |
| Al2O3 | 1.04 | 1.83 | 3.11 |
| B2O3 | 6.00 | 4.59 | 7.23 |
| CaO | 4.00 | 6.27 | 8.82 |
| Na2O | 15.20 | 15.56 | 16.24 |
| K2O | 0.26 | 0.45 | 0.71 |
| F2 | -- | 1.43 | -- |
| C(acid) | 2.06 | 1.96 | 1.35 |
| C(bio) | 2.16 | 2.14 | 1.54 |
| C(moist) | 2.89 | 2.67 | 2.11 |
| MgO | 5.23 | 4.52 | 6.36 |
| Glass | Example 3 | Example C3 |
| Element | Quantity Dissolved (ppm) | Quantity Dissolved (ppm) |
| Al | 187 | 453 |
| Ca | 2831 | 4110 |
| Mg | 854 | 938 |
| Example C5 | Example C6 | |
| SiO2 | 68.2 | 66.67 |
| Al2O3 | 2.2 | 2.25 |
| B2O3 | 5.0 | 4.78 |
| Na2O | 9.2 | 8.68 |
| K2O | -- | 0.26 |
| CaO | 11.9 | 14.77 |
| MgO | 3.5 | 3.60 |
| C(acid) | 1.85 | 1.94 |
| C(bio) | 2.03 | 2.15 |
| C(moist) | 2.54 | 2.70 |
| HTV (103 poise), est. | 1248°C (2280°F) | 1210°C (2210°F) |
| Liquidus | 1084°C (1983°F) | 1113°C (2035°F) |
| Example C7 | |
| SiO2 | 69.55 |
| Al2O3 | 0.08 |
| CaO | 7.46 |
| MgO | 4.30 |
| Na2O | 15.05 |
| K2O | 0.04 |
| B2O3 | 3.52 |
| C(acid) | 2.28 |
| C(bio) | 2.29 |
| C(moist) | 2.72 |
| HTV (103 poise) | 1095°C (2003°F) |
| Liquidus | 930°C (1706°F) |
Claims (12)
- Glass fibers exhibiting high chemical resistance, moisture resistance, and biosolubility, said glass fibers prepared from a glass composition suitable for pot and marble fiberization, said composition consisting essentially of, in mol percent:
SiO2 65.96 -- 69.7 Al2O3 0 -- 2.37 Alkaline earth metal oxide 7 -- 18 Alkali metal oxide 9 -- 20 B2O3 0 -- 7.1 - The glass fibers of claim 1 wherein said composition consists essentially of, in mol percent:
SiO2 66 -- 69.0 Al2O3 0 -- 2.2 Alkaline earth metal oxide 7 -- 16 Alkali metal oxide 9 -- 19 B2O3 0 - - 7.1 - The glass fibers of claim 2 wherein said composition consists essentially of, in mol percent:
SiO2 66 -- 68.25 Al2O3 0 -- 2.2 Alkaline earth metal oxide 7 -- 13 Alkali metal oxide 11 -- 18 B2O3 0 -- 7.1 - The glass fibers of one of claims 1 to 3 wherein ΔT is at least about 218°C (425°F).
- The glass fibers of one of claims 1 to 4 wherein said fibers have a measured biodissolution rate when fiberized by pot and marble fiberization of greater than 300 ng/cm2/hr.
- The glass fibers of claim 5 whrein ΔT is at least about 218°C (425°F) and the measured biodissolution of fibers prepared by pot and marble fiberization is greater than about 400 ng/cm2/hr.
- The glass fibers of claim 1 wherein C(acid) ≥ 2.00, C(bio) ≤ 2.23 and C(moist) ≥ 2.50.
- The glass fibers of claim 7 wherein C(acid) ≥ 2.00, C(bio) ≤ 2.20 and C(moist) ≥ 2.60.
- Flame attenuated pot and marble fiberized glass fibers, said fibers having an outer shell depleted of a position of volatile oxides, said fibers prepared from a glass composition comprising, in mol percent:
SiO2 65.96 -- 69.0 Al2O3 0 -- 2.37 Alkaline earth metal oxide 7 -- 16 Alkali metal oxide 9 -- 19 B2O3 0 -- 7.1 - The fibers of claim 9, wherein said ΔT is greater than about 204°C (400°F), said fibers exhibiting biodissolution ≥ 300 ng/cm2/hr.
- The fibers of claim 10, wherein said glass composition comprises, in mol percent:
SiO2 66 -- 68.25 Al2O3 0 -- 2.2 Alkaline earth metal oxide 7 -- 13 Alkali metal oxide 11 -- 18 B2O3 0 -- 7.1 - An acid and moisture resistant glass fiber prepared from a glass composition consisting essentially of, in mol percent:
SiO2 66.5 - 67.8 Al2O3 0.5 - 1.5 B2O3 5 - 7.0 CaO 3.0 - 7.0 MgO 3.0 - 7.0 Na2O 14.0 - 17.0 K2O 0.1 - 0.4
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US827571 | 1992-01-28 | ||
| US08/827,571 US5945360A (en) | 1997-03-28 | 1997-03-28 | Biosoluble pot and marble-derived fiberglass |
| PCT/US1998/005636 WO1998043923A1 (en) | 1997-03-28 | 1998-03-20 | Biosoluble pot and marble (flame attenuated)-derived fiberglass |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0915812A1 EP0915812A1 (en) | 1999-05-19 |
| EP0915812B1 true EP0915812B1 (en) | 2004-03-10 |
Family
ID=25249560
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP98913041A Revoked EP0915812B1 (en) | 1997-03-28 | 1998-03-20 | Biosoluble pot and marble (flame attenuated)-derived fiberglass |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US5945360A (en) |
| EP (1) | EP0915812B1 (en) |
| JP (1) | JP4718654B2 (en) |
| AU (1) | AU6768898A (en) |
| CA (1) | CA2255626C (en) |
| DE (1) | DE69822269T2 (en) |
| WO (1) | WO1998043923A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2243749A1 (en) | 2009-04-23 | 2010-10-27 | Vivoxid Oy | Resorbable and biocompatible fibre glass compositions and their uses |
| RU2751320C1 (en) * | 2020-11-23 | 2021-07-13 | Акционерное общество "НПО Стеклопластик" | Device for production of continuous high-modulus glass fibers made of thermoplastic material |
Families Citing this family (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6294491B1 (en) * | 1997-03-28 | 2001-09-25 | Johns Manville International, Inc. | Lightweight glass fiber insulation |
| CZ291441B6 (en) * | 1997-12-02 | 2003-03-12 | Rockwool International A/S | Process for forming mineral fibers |
| ATE257132T1 (en) * | 1999-04-30 | 2004-01-15 | Poliglas Sa | BIOSOLUBLE GLASS FIBER COMPOSITION FOR PRODUCING GLASS WOOLS AND THE LIKE |
| ES2163972B1 (en) * | 1999-04-30 | 2003-06-16 | Poliglas Sa | BIOSOLUBLE COMPOSITION OF GLASS FIBERS FOR THE PRODUCTION OF GLASS WOOL AND SIMILAR. |
| US6277777B1 (en) † | 1999-08-03 | 2001-08-21 | Johns Manville International, Inc. | Boron-free glass composition and filtration media |
| IT1313655B1 (en) * | 1999-09-30 | 2002-09-09 | Techint Spa | GLASS FIBER COMPOSITION. |
| AU5075901A (en) | 1999-11-03 | 2001-07-03 | Johns Manville International, Inc. | Glass composition for ultrafine fiber formation |
| DE10014678C2 (en) * | 2000-03-17 | 2002-02-07 | Siemens Ag | filter bag |
| US6828264B2 (en) * | 2001-03-28 | 2004-12-07 | Johns Manville International, Inc. | Glass compositions for ultrafine fiber formation |
| BRPI0313075A2 (en) * | 2002-07-29 | 2017-05-02 | Evanite Fiber Corp | glass compositions |
| US7144633B2 (en) * | 2002-07-29 | 2006-12-05 | Evanite Fiber Corporation | Glass compositions |
| FR2877000B1 (en) * | 2004-10-27 | 2007-08-10 | Saint Gobain Vetrotex | BIOSOLUBLES REINFORCING GLASS THREADS |
| US20060101796A1 (en) * | 2004-11-12 | 2006-05-18 | Kern Charles F | Air filtration media |
| US7648929B2 (en) * | 2004-12-30 | 2010-01-19 | Owens Corning Intellectual Capital, Llc | Low boron glass composition for loose-fill fiberglass insulation |
| GB2427191B (en) * | 2005-06-14 | 2007-06-27 | Morgan Crucible Co | Glass fibres |
| US20070016153A1 (en) * | 2005-07-13 | 2007-01-18 | Bryn Hird | Disposable absorbent articles comprising non-biopersistent inorganic vitreous microfibers |
| US20070225669A1 (en) * | 2006-03-27 | 2007-09-27 | The Procter & Gamble Company | Heterogeneous absorbent cores |
| US8198505B2 (en) * | 2006-07-12 | 2012-06-12 | The Procter & Gamble Company | Disposable absorbent articles comprising non-biopersistent inorganic vitreous microfibers |
| US7763558B2 (en) * | 2006-12-27 | 2010-07-27 | Johns Manville | Glass compositions for fiber formation |
| US8142535B2 (en) * | 2008-08-05 | 2012-03-27 | Johns Manville | High dust holding capacity filter media |
| US8057583B2 (en) * | 2008-08-07 | 2011-11-15 | Johns Manville | Filter media including silicone and/or wax additive(s) |
| PL2243500T3 (en) | 2009-04-23 | 2012-06-29 | Purac Biochem Bv | Biocompatible composite and its use |
| WO2010125981A1 (en) * | 2009-04-28 | 2010-11-04 | 旭硝子株式会社 | Glass plate for substrate |
| JP2014532525A (en) | 2011-11-09 | 2014-12-08 | ザ プロクター アンド ギャンブルカンパニー | Absorbent article with double core |
| US9267238B2 (en) | 2012-07-25 | 2016-02-23 | Johns Manville | Glass fiber reinforced facer mat |
| GB2528004A (en) | 2013-05-08 | 2016-01-06 | Procter & Gamble | Absorbent article with dual core |
| CA2927857C (en) | 2014-08-14 | 2022-12-06 | Hdk Industries, Inc. | Apparatus and method for filtration efficiency improvements in fibrous filter media |
| US10729600B2 (en) | 2015-06-30 | 2020-08-04 | The Procter & Gamble Company | Absorbent structure |
| US10003056B2 (en) | 2015-09-30 | 2018-06-19 | Johns Manville | Battery containing acid resistant nonwoven fiber mat with biosoluble microfibers |
| RU2697170C1 (en) | 2015-11-04 | 2019-08-12 | Дзе Проктер Энд Гэмбл Компани | Absorbent structure |
| US11173078B2 (en) | 2015-11-04 | 2021-11-16 | The Procter & Gamble Company | Absorbent structure |
| CA3004313A1 (en) | 2015-11-04 | 2017-05-11 | The Procter & Gamble Company | Absorbent structure |
| FR3053965B1 (en) | 2016-07-13 | 2018-08-17 | Saint-Gobain Isover | GLASS FIBERS |
Family Cites Families (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2692654A (en) * | 1953-03-12 | 1954-10-26 | Pittsburgh Plate Glass Co | Impingement-strainer combination air filter |
| US2823117A (en) * | 1953-11-23 | 1958-02-11 | L O F Glass Fibers Inc | Glass paper-calcium silicate |
| US3663192A (en) * | 1969-11-28 | 1972-05-16 | Johns Manville | Glass melting pot |
| US3811853A (en) * | 1972-05-01 | 1974-05-21 | Corning Glass Works | Degradable glass suitable for containers |
| US3840359A (en) * | 1972-05-18 | 1974-10-08 | Philadelphia Quartz Co | Preparation of hydrated alkali metal silicate flakes |
| US3972694A (en) * | 1974-11-14 | 1976-08-03 | Whatman Reeve Angel Limited | Filter tube |
| FR2552075B1 (en) * | 1983-09-19 | 1986-08-14 | Saint Gobain Isover | GLASS FIBERS AND COMPOSITION SUITABLE FOR THEIR MANUFACTURE |
| US4510252A (en) * | 1983-11-18 | 1985-04-09 | Owens-Corning Fiberglas Corporation | Easily formed chemically resistant glass fibers |
| US4542106A (en) * | 1983-12-19 | 1985-09-17 | Ppg Industries, Inc. | Fiber glass composition |
| US5332699A (en) * | 1986-02-20 | 1994-07-26 | Manville Corp | Inorganic fiber composition |
| US5332698A (en) * | 1989-05-25 | 1994-07-26 | Bayer Aktiengesellschaft | Glass fibres with improved biological compatibility |
| US5250488A (en) * | 1989-08-11 | 1993-10-05 | Sylvie Thelohan | Mineral fibers decomposable in a physiological medium |
| AU630484B2 (en) * | 1989-08-11 | 1992-10-29 | Isover Saint-Gobain | Glass fibres capable of decomposing in a physiological medium |
| US5055428A (en) * | 1990-09-26 | 1991-10-08 | Owens-Corning Fiberglass Corporation | Glass fiber compositions |
| US5401693A (en) * | 1992-09-18 | 1995-03-28 | Schuller International, Inc. | Glass fiber composition with improved biosolubility |
| JPH06116114A (en) * | 1992-10-09 | 1994-04-26 | Nikon Corp | Bone filler |
| DK156692D0 (en) * | 1992-12-29 | 1992-12-29 | Rockwool Int | MINERAL FIBER PRODUCT |
| US5472467A (en) * | 1994-03-14 | 1995-12-05 | Pfeffer; Jack R. | Self-supporting filter composite |
| US5634954A (en) * | 1994-03-30 | 1997-06-03 | Schuller International, Inc. | Fibrous filter media |
| US5591453A (en) * | 1994-07-27 | 1997-01-07 | The Trustees Of The University Of Pennsylvania | Incorporation of biologically active molecules into bioactive glasses |
| US5728187A (en) * | 1996-02-16 | 1998-03-17 | Schuller International, Inc. | Air filtration media |
| US5785725A (en) * | 1997-04-14 | 1998-07-28 | Johns Manville International, Inc. | Polymeric fiber and glass fiber composite filter media |
-
1997
- 1997-03-28 US US08/827,571 patent/US5945360A/en not_active Expired - Lifetime
-
1998
- 1998-03-20 CA CA002255626A patent/CA2255626C/en not_active Expired - Lifetime
- 1998-03-20 DE DE69822269T patent/DE69822269T2/en not_active Expired - Lifetime
- 1998-03-20 AU AU67688/98A patent/AU6768898A/en not_active Abandoned
- 1998-03-20 EP EP98913041A patent/EP0915812B1/en not_active Revoked
- 1998-03-20 JP JP53991198A patent/JP4718654B2/en not_active Expired - Lifetime
- 1998-03-20 WO PCT/US1998/005636 patent/WO1998043923A1/en not_active Ceased
-
1999
- 1999-07-08 US US09/349,552 patent/US6261335B1/en not_active Expired - Lifetime
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2243749A1 (en) | 2009-04-23 | 2010-10-27 | Vivoxid Oy | Resorbable and biocompatible fibre glass compositions and their uses |
| WO2010122019A1 (en) | 2009-04-23 | 2010-10-28 | Vivoxid Oy | Resorbable and biocompatible fibre glass compositions and their uses |
| RU2751320C1 (en) * | 2020-11-23 | 2021-07-13 | Акционерное общество "НПО Стеклопластик" | Device for production of continuous high-modulus glass fibers made of thermoplastic material |
Also Published As
| Publication number | Publication date |
|---|---|
| DE69822269D1 (en) | 2004-04-15 |
| US5945360A (en) | 1999-08-31 |
| WO1998043923A1 (en) | 1998-10-08 |
| JP2000511508A (en) | 2000-09-05 |
| EP0915812A1 (en) | 1999-05-19 |
| AU6768898A (en) | 1998-10-22 |
| DE69822269T2 (en) | 2005-04-21 |
| JP4718654B2 (en) | 2011-07-06 |
| CA2255626C (en) | 2007-06-12 |
| US6261335B1 (en) | 2001-07-17 |
| CA2255626A1 (en) | 1998-10-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0915812B1 (en) | Biosoluble pot and marble (flame attenuated)-derived fiberglass | |
| AU2002324024C9 (en) | Biosoluble ceramic fiber composition with improved solubility in a physiological saline solution for a high temperature insulation material | |
| EP0918734B1 (en) | Glass compositions for high thermal insulation efficiency glass fibers | |
| EP1235755B1 (en) | Glass composition for ultrafine fiber formation | |
| US20030015003A1 (en) | Low temperature glass for insulation fiber | |
| EP1979284B1 (en) | A biodegradable ceramic fiber composition for a heat insulating material | |
| KR101223675B1 (en) | Saline soluble ceramic fiber composition | |
| AU741801B2 (en) | Artificial mineral wool composition | |
| EP1048625A1 (en) | Biosoluble composition of glass fibres for the production of glass wools and the like | |
| US6794321B2 (en) | Glass compositions for ultrafine fiber information | |
| US6399525B2 (en) | Flame attenuated fiberglass | |
| US5932347A (en) | Mineral fiber compositions | |
| US6828264B2 (en) | Glass compositions for ultrafine fiber formation | |
| CA2575900C (en) | Biosoluble pot and marble-derived fiberglass | |
| WO1998018618A1 (en) | Mineral fiber compositions | |
| MXPA98009902A (en) | Biosoluble pot and marble (flame attenuated)-derived fiberglass | |
| KR100789469B1 (en) | Composition of biosoluble glass fiber | |
| KR20250172151A (en) | Composition for ceramic fiber and ceramic fiber manufactured therefrom | |
| WO1998040322A1 (en) | Dual glass compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 19990312 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): BE DE FR GB IT NL SE |
|
| 17Q | First examination report despatched |
Effective date: 20001205 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): BE DE FR GB IT NL SE |
|
| REF | Corresponds to: |
Ref document number: 69822269 Country of ref document: DE Date of ref document: 20040415 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| ET | Fr: translation filed | ||
| PLBQ | Unpublished change to opponent data |
Free format text: ORIGINAL CODE: EPIDOS OPPO |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: SAINT-GOBAIN ISOVER Effective date: 20041208 |
|
| NLR1 | Nl: opposition has been filed with the epo |
Opponent name: SAINT-GOBAIN ISOVER |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| PLAB | Opposition data, opponent's data or that of the opponent's representative modified |
Free format text: ORIGINAL CODE: 0009299OPPO |
|
| R26 | Opposition filed (corrected) |
Opponent name: SAINT-GOBAIN ISOVER Effective date: 20041208 |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| NLR1 | Nl: opposition has been filed with the epo |
Opponent name: SAINT-GOBAIN ISOVER |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20110331 Year of fee payment: 14 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20110325 Year of fee payment: 14 Ref country code: BE Payment date: 20110330 Year of fee payment: 14 Ref country code: DE Payment date: 20110329 Year of fee payment: 14 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R103 Ref document number: 69822269 Country of ref document: DE Ref country code: DE Ref legal event code: R064 Ref document number: 69822269 Country of ref document: DE |
|
| APBU | Appeal procedure closed |
Free format text: ORIGINAL CODE: EPIDOSNNOA9O |
|
| RDAF | Communication despatched that patent is revoked |
Free format text: ORIGINAL CODE: EPIDOSNREV1 |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| 27W | Patent revoked |
Effective date: 20110819 |
|
| GBPR | Gb: patent revoked under art. 102 of the ep convention designating the uk as contracting state |
Effective date: 20110819 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R107 Ref document number: 69822269 Country of ref document: DE Effective date: 20120412 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: ECNC |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20120327 Year of fee payment: 15 Ref country code: SE Payment date: 20120328 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20120329 Year of fee payment: 15 |